Sculpting Marble In Nature - The Effects Of Coccolith Formation
The world and its living inhabitants are connected, with geophysical processes ubiquitously affecting natural ecosystems and vice versa. Over the span of millennia and eons, the ponderous drift of tectonic plates might alter the size and location of continents around the oceans to separate populations of the same species from each other, causing them to evolve independently from each other. Like this, through allopatric speciation, new species are created. Likewise, the absorbance, decomposition and waste of biological organisms continuously ascertains that elements in nature remain in circulation, simultaneously affecting the mineral compositions of rocky reservoirs inside biodiverse soil and even mountains.
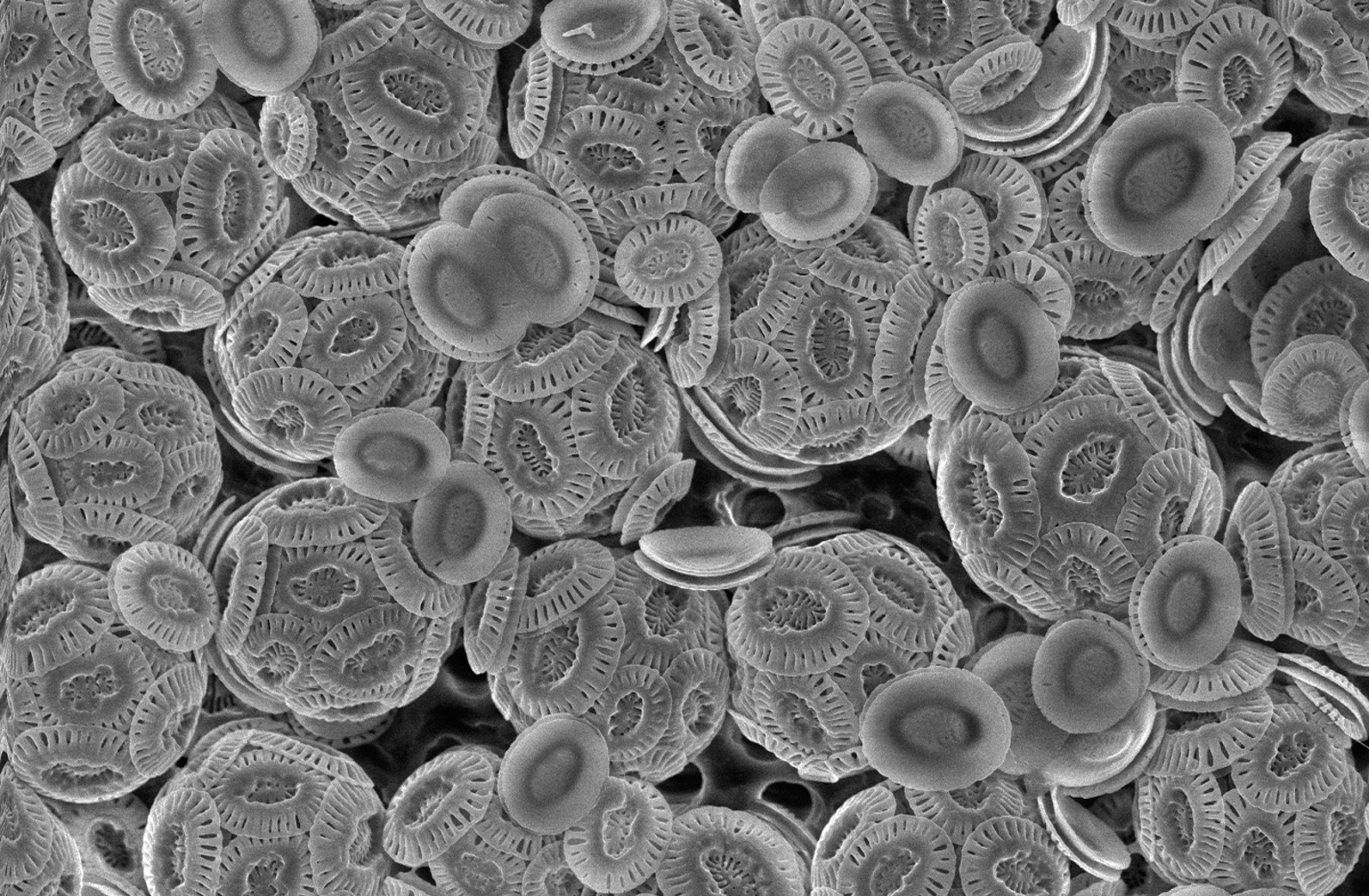
It is easy to come by such relationships in nature. Ecosystems, after all, are not whole without the abiotic factors that support their encompassing communities. Some creatures, however, are more visibly associated with the Earth than others - and none showcase it better than the coccolithophores. Belonging to the Kingdom Protista and the Order Haptophyta, these bumbling unicellular phytoplankton (otherwise known as microalgae) are exemplar models of the extent that biological process can influence our planet's geography. They are also miniscule, measuring up to some 75 micrometers in diameter, if that. With this in mind - how are coccolithophores able to build underwater marble mountains out of thin air?
The Diversity Of Marble Shells
'Out of thin air' is admittedly not quite right. Usually surrounded by large quantities of dissolved calcium and low concentrations of magnesium ions, coccolithophores may be found floating on the topmost layer of the ocean surface, spread throughout the water to make the most efficient use of sunlight for photosynthesis. More renown, though, is their ability to absorb dissolved calcium salts and carbon dioxide to create calcium carbonate (i.e., calcite, or marble) shells - the so-called coccoliths.
Composing beautifully ornamented calcite surfaces around them, coccolith formation is known to be one of the largest contributors to marine carbon supplies, as well as a buffer to the increasing acidity of the oceans. It also makes their makers appear white and foamy as a population. Accordingly, if you've ever wondered what the white foam on beach water is, here you go. It is the shells of the humble coccolithophore and its billions of siblings and colleagues, all visiting to say hello. This kind of biomineralization is also interestingly common in nature. Snails, too, make use of calcium carbonate to construct their shells, with pearl oysters having the rather profitable tendency to form white pearls as salt reservoirs in their enclosures.

In the case of the everyday coccolithophore, its shell actually has the unique property of being either spherically or cylindrically symmetrical, featuring only small pores that break up the encompassing coccosphere to allow the diffusion of molecules into and out of the phytoplankton. In light of this, there are two main classifications that scientists give them: heterococcoliths, formed from multiple repeats of elaborately structured crystal patterns; and holococcoliths, which are generally simpler arrays of regularly interspaced calcite grains. At first, oceanographers believed that different types of coccoliths were produced and secreted by different species. However, the last two decades have largely disproved this; both types of marble shells have been observed on organisms of the same species, further displaying astonishing variations of patterns between individuals. Scientists have since discovered that the type of coccolith secreted by a coccolithophore depends on both its environment - particularly on the surrounding's pH level, calcium ion concentration and dissolved carbon dioxide - and its stage in life.
Coccolithophores exhibit a heteromorphic life cycle. Essentially, this means that cells appear to have different amounts of genetic material in separate periods of their life, so that one time they might be haploid (containing only one set of chromosomes) and another time when they will be diploid (with doubled chromosomes that form homologous pairs). This phenomenon occurs due to the asexual reproduction of coccolithophores, dividing via both meiosis and mitosis to obtain haploid cells before reverting back to diploidy via a complex metabolic process referred to as syngamy. The details of these mechanisms are inconsequential to the current matter, though - so just know that holococcoliths occur mostly in haploid coccolithophores, while heterococcoliths are much more common in their diploid counterparts. Currently, the evolutionary significance behind these changes is somewhat poorly understood, and so more research is required to appreciate them.
Albeit, one common attribute between the different shells is their manner of secretion. Taking place through biochemical calcification pathways inside the cell, several models indicate that coccoliths do not only offer physical protection to their phytoplankton, but are also involved in the chemical regulation of their immediate surroundings, their resistance to harmful radiation and even their metabolic growth.
Cellular Calcification
Interestingly, coccolith formation apparently takes place from the inside-out. Calcium and bicarbonate ions (the latter of which are the product of dissolved carbon dioxide) are at first transported into the cell through gated protein channels integrated in its membrane. Being the most energy-consuming step in the mechanism (especially in concern with calcium transport, which goes against both its concentration and electrical gradients), the rate at which this happens directly correlates to how rapidly coccoliths are generated. Mineralisation then occurs near the Golgi apparatus, a large eukaryotic organelle deeply involved in the packaging and vesicular trafficking of compounds around a cell. Here, the Golgi directs calcium-binding proteins (CAPs) that bind to newly formed calcite in the cytoplasm, henceforth catalysing calcite crystallisation into coccolith plates. (Otherwise known as nucleation, this type of reaction happens amongst a number of kindred 'build-a-block' processes in the cell, including actin polymerisation, amongst others.)

Like a canvas ready to be painted on, CAPs allow complex sugars to join and alter the shape of the constructed calcite crystals, ultimately creating the artistic imprints that we saw before. Perfectly decorated, the finished plates are then placed inside secretory vesicles before being sent out into the outside world (via a process known as exocytosis, wherein the vesicles fuse with the cell membrane to release their contents). Now, notice: since calcite is nucleated inside the cell, it means that fresh new coccoliths will be found underneath their older variants. As such, in order to prevent from clogging itself up with its shells, coccolithophores have to control its rate of calcification so according to the state of its outer shells in time. Since this is proportional to factors like the pH of the surroundings - which, if low, steadily damages and dissolves calcium carbonate - this regulation plays a role in the cell's response mechanisms to its environment. Not to mention that this becomes especially important when you realise that the cells have to secrete hydrogen ions to allow biomineralisation to take place, indirectly decreasing the pH. But I digress.
Coccoliths have further been observed to be significant factors in physical protection against high-energy radiation (e.g., ultraviolet light from the sun), viral or bacterial infections (which can be particularly harmful to them, given that coccolithophores have no immune response), and even against larger microbiotic predators in their ecosystem. Species such as the Rhabdosphaera clavigera, found in the upper layer of the ocean, have developed spikes or elongated coccoliths to deter both specific and non-specific grazers like stentors in the water. I say this, but it probably does not work as you might first think. A cell of that size won't really be bothered by a smaller particle poking it in the metaphorical face. It'll just eat it. Given this fact, researchers believe that the coccolith extensions could instead act as 'junk food' that must be peeled off before a grazer can reach the more precious organic material within. Naturally, this process takes a lot of energy and effort, and hence would disrupt the average grazer from constantly feeding on entire populations of coccolithophores nearby.

Another major use of the coccolith, marine biologists theorise, is its way of filtering in visible light to use for photosynthesis (where carbon dioxide is fixated to produce sugars and other important biomolecules in the cell). Being the most relevant in plankton from the deep ocean - where light is understandably quite poor - coccoliths on such species are known to be more layered than others. The shape allows light rays to be evenly dispersed amongst chloroplasts in the cell, ultimately achieving a far greater efficiency than would otherwise be possible. Similarly, calcification is able to concentrate dissolved carbon dioxide by acidifying the surrounding water. It is a cruel irony that the ocean acidification of global warming has apparently been seen to exponentially increase the number of coccolithophore blooms in the ocean.
Having said all of this, you may now understand how coccolithophores have become some of the most succesful organisms on Earth, literally inhabiting every ocean (except for the Artic) since the time of the dinosaurs. Luckily, they also seem to be great housekeepers - and they could very well be crucial in stopping the terrestial effects of climate change.
Altering The Sky
One popular theory is that blooms of these cells could trap carbon dioxide out of the atmosphere in their shells, thereby sequestering them in the ocean floor after sinking. Even though coccolith formation does lower the pH some, the net effect of trapping greenhouse gases both reduces global warming and balances out the pH change of the oceans. Over time, once coccolithophore blooms have declined (i.e., when carbon dioxide and acidification have decreased), pH would be further stabilised to regular levels to support a more diverse aquatic ecosystem.
There are plenty of studies demonstrating this effect, praising the capability of coccolithophores to alter the climate and provide wonders to life underwater. Of course, a few others have claimed otherwise, providing evidence that coccoliths contribute to the air-polluting effect of sea spray aerosols. Whether this is very significant is up for debate. Either way, I believe that it is still quite lovely to have the critters around - because how often do you really see artists quite like them?
References
- Müller, M. N. (2019). On the Genesis and Function of Coccolithophore Calcification. Frontiers in Marine Science. Retrieved from https://doi.org/10.3389/fmars.2019.00049
- Taylor, A. R. (2007). Dynamics of formation and secretion of heterococcoliths by Coccolithus pelagicus ssp. braarudii. European Journal of Phycology 42(2). Retrieved from https://doi.org/10.1080/09670260601159346
- Monteiro, F. M., et al (2016). Why marine phytoplankton calcify. Science Advances 2(7):e1501822. Retrieved from https://doi.org/10.1126/sciadv.1501822
- Sanders, R., et al. (2010). Does a ballast effect occur in the surface ocean? Geophysical Research Letters 37:L08602. Retrieved from https://doi.org/10.1029/2010GL042574
- Erba, E. (2006). The first 150 million years history of calcareous nannoplankton: Biosphere–geosphere interactions. Palaeogeography, Palaeoclimatology, Palaeoecology 232:237-250. Retrieved from https://doi-org.iclibezp1.cc.ic.ac.uk/10.1016/j.palaeo.2005.09.013
- Trainic, M., et al (2018). Infection Dynamics of a Bloom-Forming Alga and Its Virus Determine Airborne Coccolith Emission from Seawater. Cell 6:327-335. Retrieved from https://doi.org/10.1016/j.isci.2018.07.017
